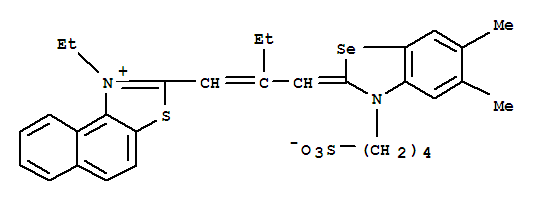
2-[2-[[5,6-dimethyl-3-(4-sulphonatobutyl)-3H-benzoselenazol-2-ylidene]methyl]-1-butenyl]-1-ethylnaphtho[1,2-d]thiazolium
Basic Information
- CAS No.: 94213-13-5
Physical properties
Product Description
Reputable supplier selling 2-[2-[[5,6-dimethyl-3-(4-sulphonatobutyl)-3H-benzoselenazol-2-ylidene]methyl]-1-butenyl]-1-ethylnaphtho[1,2-d]thiazolium 94213-13-5 with stock
- Molecular Formula: C31H34 N2 O3 S2 Se
- Molecular Weight: 625.70326
- Boiling Point: °Cat760mmHg
- Flash Point: °C
- Density: g/cm3
2-[2-[[5,6-dimethyl-3-(4-sulphonatobutyl)-3H-benzoselenazol-2-ylidene]methyl]-1-butenyl]-1-ethylnaphtho[1,2-d]thiazolium(Cas 94213-13-5) Usage
|
Type of compound |
Complex organic compound |
|
Elements present |
Selenium, sulphur, and carbon |
|
Structure |
Long hydrocarbon chain, sulfonate group, benzothiazolium ring, and selenium atom |
|
Usage in research |
Dye or fluorescent probe |
|
Application |
Biological research, analytical chemistry, and photodynamic therapy |
|
Property |
Ability to bind to certain biological structures and emit detectable light |
|
Function |
Chemical indicator for detecting specific ions or molecules |
|
Therapeutic potential |
Generation of reactive oxygen species for selectively destroying cancer cells |
|
General Description |
The chemical "2-[2-[[5,6-dimethyl-3-(4-sulphonatobutyl)-3H-benzoselenazol-2-ylidene]methyl]-1-butenyl]-1-ethylnaphtho[1,2-d]thiazolium" is a complex organic compound containing selenium, sulphur, and naphthalene rings. It has a long hydrocarbon chain with a sulfonate group attached, and a benzothiazolium ring containing a selenium atom. 2-[2-[[5,6-dimethyl-3-(4-sulphonatobutyl)-3H-benzoselenazol-2-ylidene]methyl]-1-butenyl]-1-ethylnaphtho[1,2-d]thiazolium is often used as a dye or fluorescent probe in biological research, due to its ability to bind to certain biological structures and emit detectable light. It is also used as a chemical indicator in analytical chemistry, to detect the presence of specific ions or molecules. Additionally, it has potential applications in photodynamic therapy, due to its ability to generate reactive oxygen species upon activation, which can be used to selectively destroy cancer cells. |
InChI:InChI=1/C31H34N2O3S2Se/c1-5-23(19-29-32(6-2)31-25-12-8-7-11-24(25)13-14-27(31)37-29)20-30-33(15-9-10-16-38(34,35)36)26-17-21(3)22(4)18-28(26)39-30/h7-8,11-14,17-20H,5-6,9-10,15-16H2,1-4H3