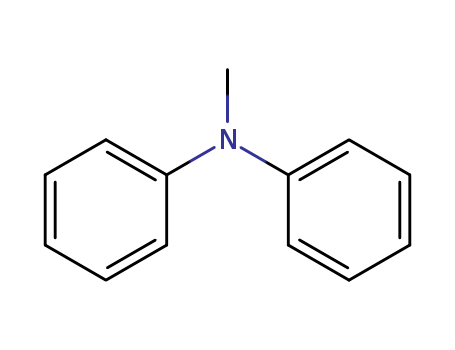
Your location:Home > Agrochemicals >N-Methyldiphenylamine
N-Methyldiphenylamine
Basic Information
- CAS No.: 552-82-9
Physical properties
Product Description
Factory Sells Best Quality N-Methyldiphenylamine 552-82-9 with GMP standards
- Molecular Formula: C13H13 N
- Molecular Weight: 183.253
- Appearance/Colour: yellow oil-like liquid
- Vapor Pressure: 0.00143mmHg at 25°C
- Melting Point: -7.6 ºC
- Refractive Index: n20/D 1.623(lit.)
- Boiling Point: 296-297 ºC
- PKA: 1.00±0.20(Predicted)
- Flash Point: 116.6 ºC
- PSA: 3.24000
- Density: 1.05
- LogP: 3.45450
N-Methyldiphenylamine(Cas 552-82-9) Usage
|
Hazard |
Toxic by ingestion. |
|
General Description |
N-Methyldiphenylamine is an aromatic tertiary amine. It undergoes transformation to N-methylcarbazole (C) via a photochemical reaction. |
InChI:InChI=1/C13H13N/c1-14(12-8-4-2-5-9-12)13-10-6-3-7-11-13/h2-11H,1H3/p+1
552-82-9 Relevant articles
Three-Component Coupling Involving Arynes, Aromatic Tertiary Amines, and Aldehydes via Aryl-Aryl Amino Group Migration
Bhojgude, Sachin Suresh,Baviskar, Dnyaneshwar R.,Gonnade, Rajesh G.,Biju, Akkattu T.
, p. 6270 - 6273 (2015)
The transition-metal-free multicomponent...
Borane-Trimethylamine Complex as a Reducing Agent for Selective Methylation and Formylation of Amines with CO2
Zhang, Yanmeng,Zhang, He,Gao, Ke
supporting information, p. 8282 - 8286 (2021/10/25)
We report herein that a borane-trimethyl...
Effect of Precatalyst Oxidation State in C-N Cross-Couplings with 2-Phosphinoimidazole-Derived Bimetallic Pd(I) and Pd(II) Complexes
Martinez, Erin E.,Moreno, Mariur Rodriguez,Barksdale, Caleb A.,Michaelis, David J.
supporting information, p. 2763 - 2767 (2021/08/27)
We report the catalytic activity of two ...
Mesoionic N-heterocyclic olefin catalysed reductive functionalization of CO2for consecutiveN-methylation of amines
Das, Arpan,Maji, Subir,Mandal, Swadhin K.
, p. 12174 - 12180 (2021/09/28)
A mesoionic N-heterocyclic olefin (mNHO)...
Univariate classification of phosphine ligation state and reactivity in cross-coupling catalysis
Newman-Stonebraker, Samuel H.,Smith, Sleight R.,Borowski,Peters, Ellyn,Gensch, Tobias,Johnson, Heather C.,Sigman, Matthew S.,Doyle, Abigail G.
, p. 301 - 308 (2021/10/22)
Chemists often use statistical analysis ...
552-82-9 Process route
-
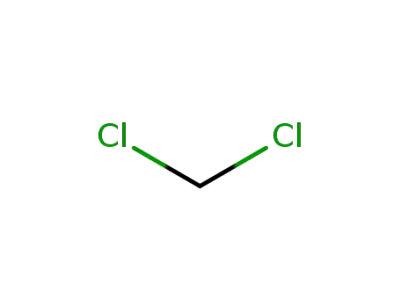
-
75-09-2
dichloromethane

-
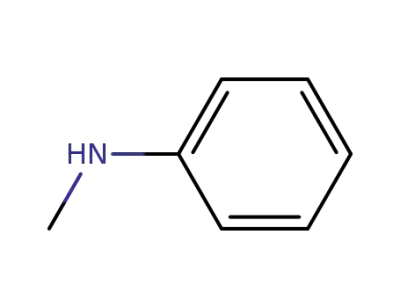
-
100-61-8
N-methylaniline

-
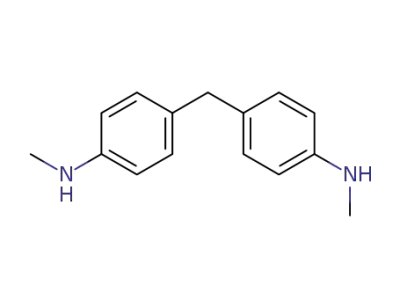
-
1807-55-2
4,4'-methylenebis(N-methylaniline)

-
-
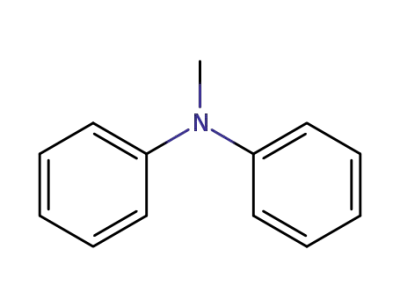
552-82-9
benzhydrylidene(methyl)amine

-
-
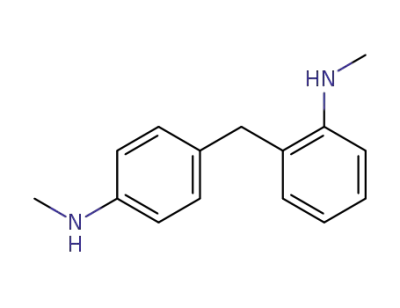
4073-99-8
N,N'-dimethyl-2,4'-diaminodiphenylmethane

-
-
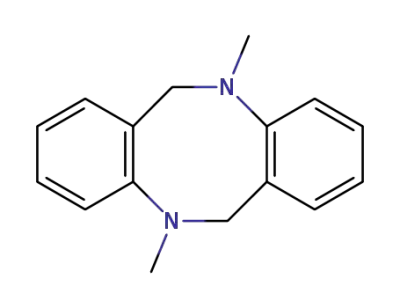
7165-20-0
1,2,5,6-tetrahydro-1,5-dimethyl-3,4,7,8-dipheno-1,5-diazocine
| Conditions | Yield |
|---|---|
|
In
benzene;
at 15 ℃;
for 48h;
Further byproducts given;
Irradiation;
|
12.7%
|
|
at 15 ℃;
for 48h;
Further byproducts given;
Irradiation;
|
6.3%
4.1% 8.1% |
-
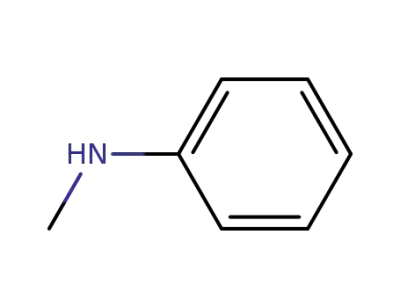
-
100-61-8
N-methylaniline

-

-
1807-55-2
4,4'-methylenebis(N-methylaniline)

-

-
552-82-9
benzhydrylidene(methyl)amine

-

-
4073-99-8
N,N'-dimethyl-2,4'-diaminodiphenylmethane

-

-
7165-20-0
1,2,5,6-tetrahydro-1,5-dimethyl-3,4,7,8-dipheno-1,5-diazocine
| Conditions | Yield |
|---|---|
|
With
dichloromethane;
at 15 ℃;
for 48h;
Further byproducts given;
Irradiation;
|
6.3%
4.1% 8.1% |
552-82-9 Upstream products
-
67-56-1
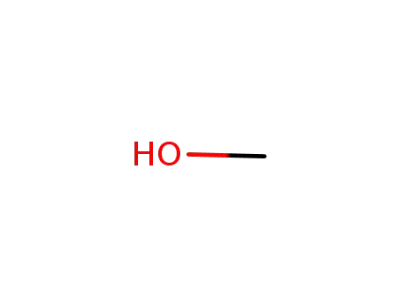
methanol
-
537-67-7
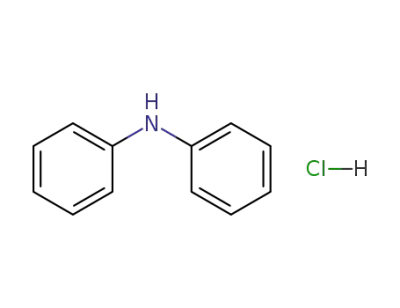
diphenylamine hydrochloride
-
607-00-1
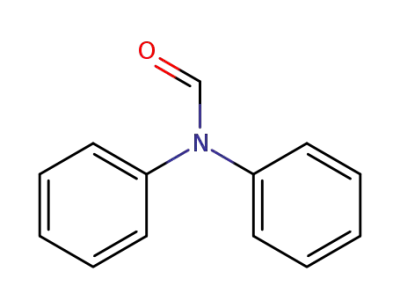
diphenylformamide
-
603-52-1
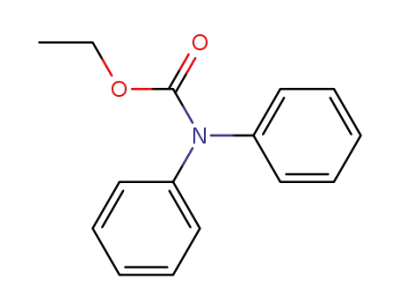
ethyl diphenylcarbamate
552-82-9 Downstream products
-
382165-80-2
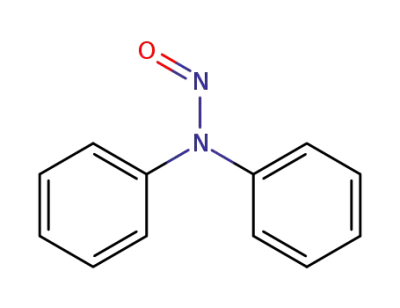
N-nitrosodiphenylamine
-
68258-04-8
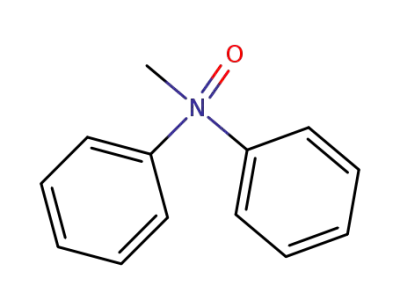
N-methyl-N-phenylaniline N-oxide
-
20349-66-0
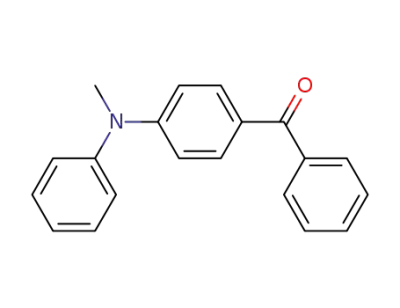
(4‐(methyl(phenyl)amino)phenyl)(phenyl)methanone
-
73323-82-7
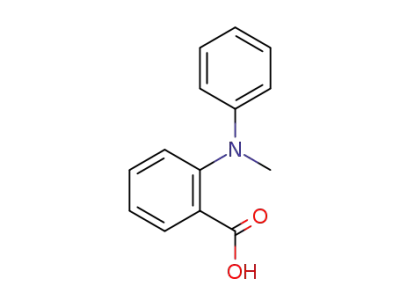
2-(N-Methyl-N-phenylamino)-benzoic acid