Your location:Home > Pharmaceutical >oxyphenisatine di(acetate)
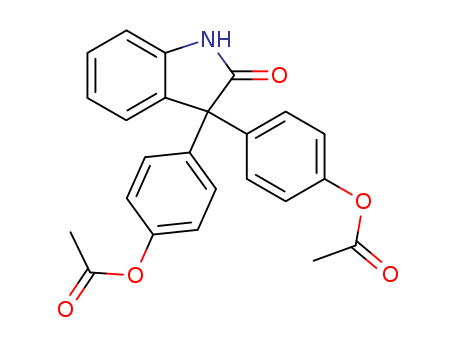
oxyphenisatine di(acetate)
Basic Information
- CAS No.: 115-33-3
Physical properties
Product Description
Factory sells oxyphenisatine di(acetate) 115-33-3 with sufficient production capacity
- Molecular Formula: C24H19 N O5
- Molecular Weight: 401.419
- Vapor Pressure: 7.5E-13mmHg at 25°C
- Melting Point: 244℃
- Boiling Point: 566.5 °C at 760 mmHg
- PKA: 13.55±0.40(Predicted)
- Flash Point: 296.4 °C
- PSA: 81.70000
- Density: 1.282 g/cm3
- LogP: 3.96170
oxyphenisatine di(acetate)(Cas 115-33-3) Usage
|
Manufacturing Process |
235 gravimetrical parts of acetic acid anhydride (90%) are poured over 106 gravimetrical parts of diphenolisatin (Berichte der Deutschen Chemischen Gesselschsft, 18, 1885, p. 2641) and the mixture is heated on the water-bath while stirring. The solid starting material temporarily dissolves almost entirely and shortly afterwards the reaction product turns into a crystalline paste. In order to complete the reaction the heating on the water-bath is continued for a short time and then the whole is left to get cold. The reaction product may, for instance, be separated in the following manner: To the cold reaction mixture is gradually added about the same volumetrical quantity of alcohol; in this manner the excess of acetic acid anhydride is destroyed and the paste becomes thinner. Then the fluid is drawn off and the product washed with alcohol. For complete cleansing another extraction is made with warm alcohol and the product crystallized, for instance, from 10 parts of acetic acid. The product represents a light, fine crystalline powder, which is difficultly soluble or even insoluble in the usual organic solvents. Its melting point lies at 242°C. |
|
Therapeutic Function |
Laxative |
|
World Health Organization (WHO) |
Oxyphenisatine acetate was widely used as a laxative after its cathartic activity was first described in 1925. In 1969 its use was first associated with cases of acute and chronic liver disease. This association is considered by some, but not all, national drug regulatory authorities to warrant the withdrawal from the market of preparations containing oxyphenisatine and its derivatives. |
|
Brand name |
Isocrin (Parke-Davis); Lavema (Sterling Winthrop);Acelax;Acetalax;Alophen pills;Ameiax;Api-slender;Artzone;Belloform;Biivectan;Bisflatan;Boxogetten;Brocatine;Buta pirone;Buteril;Butofen;Butolfen;Bydolax;Chlofel;Chur-lax;Ciracen;Cirotex;Cirotyl;Contax;Critex;Curolax;Darmoletten;Deililax;Dialose plus;Diasatin;Ditinil;Ditmedol;Eulaxin;Evac-u-lax;Ex-lax pills;Febutolo;Fibutrox;Fim-a-mint;Fin-a-mint gum;Fisiolax;Flib 518;Inlax;Ipebutona;Isaaxan;Isaphenyn;Izaman;Laveman;Laxan-vomoxin;Laxaseptol;Laxem;Laxnormal;Laxocoleva;Laxon;Laxos;Laxyl;Med-laxan;Menabil complex;Muxol;Naleran;Neocervulax;Neo-favmadol;Neo-soldana;Nourilax;Nurilaksi;Obstilax;Otone;Oxybutazone;Phlogase;Phlogistol;Phlogont;Potsilo;Promassoletten;Prusol;Rheumapax;Rivolax;Schokilax;Syndian;Tanderil-alka;Tete-lax;Validil. |
InChI:InChI=1/C24H19NO5/c1-15(26)29-19-11-7-17(8-12-19)24(18-9-13-20(14-10-18)30-16(2)27)21-5-3-4-6-22(21)25-23(24)28/h3-14H,1-2H3,(H,25,28)
115-33-3 Process route
-
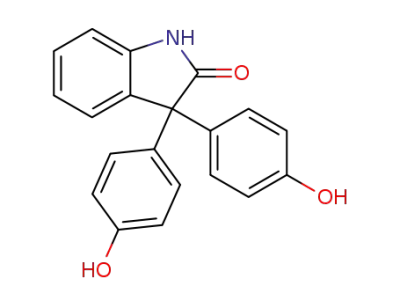
-
125-13-3
oxyphenisatine

-
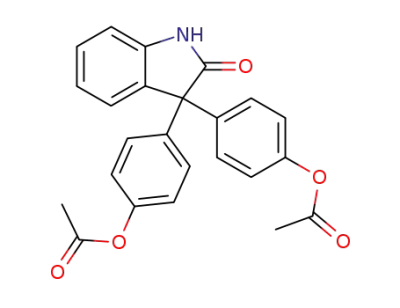
-
115-33-3
Bisatin
| Conditions | Yield |
|---|---|
|
With
sodium hydroxide; water; acetic anhydride;
at -5 - 0 ℃;
|
|
|
With
pyridine; benzoyl chloride; acetic acid;
at 0 ℃;
|
|
|
With
PPA; acetic acid;
|
-
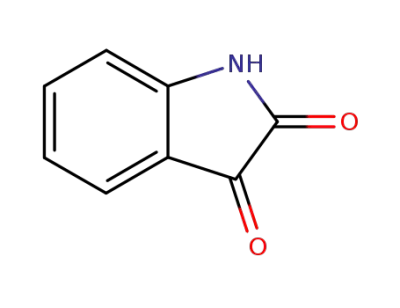
-
91-56-5,1186480-61-4,84788-92-1
indole-2,3-dione

-
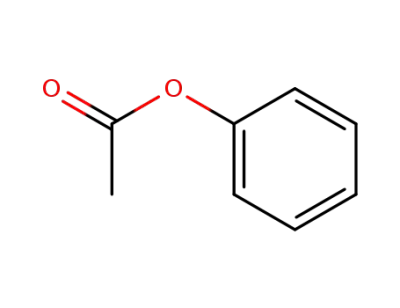
-
122-79-2,34474-31-2
Phenyl acetate

-

-
115-33-3
Bisatin
| Conditions | Yield |
|---|---|
|
With
trichlorophosphate;
|
|
|
With
sulfuric acid; phosphorus pentoxide;
at 85 - 90 ℃;
|
115-33-3 Upstream products
-
39033-45-9
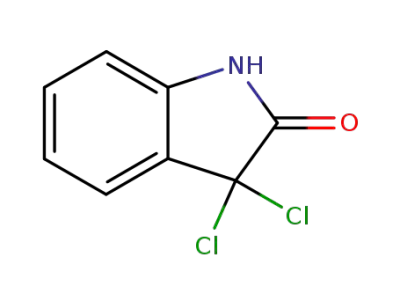
3,3-dichloro-2,3-dihydro-1H-indol-2-one
-
122-79-2

Phenyl acetate
-
125-13-3

oxyphenisatine
-
108-24-7
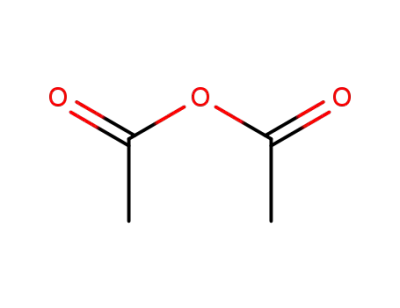
acetic anhydride